Research
Eukaryotic organisms have evolved in a bacterial world. As such, they enter into symbiotic relationships with their bacterial partners and the complexity of their reciprocal dialogue is only now starting to be unraveled. Together with nutrition, microbiota is one of the key forces impacting on host physiological status. The group of Integrative Physiology of Gnotobionts uses the germ-free mouse model to study the microbiota, specific bacterial strains and bacterial antigenic components impact on the physiological status of the eukaryotic host in two main research lines 1) host juvenile growth and 2) prevention/therapy of allergic sensitization and allergy development.
Host juvenile growth
 We have shown for the first time that microbiota is necessary to sustain optimal weight gain and linear growth of infant mice when fed a standard diet or a nutritionally depleted isocaloric diet (Schwarzer et al., 2016Makki K, Storelli G, Machuca-Gayet I, Srutkova D, Hermanova P, Martino ME, Balmand S, Hudcovic T, Heddi A et al
We have shown for the first time that microbiota is necessary to sustain optimal weight gain and linear growth of infant mice when fed a standard diet or a nutritionally depleted isocaloric diet (Schwarzer et al., 2016Makki K, Storelli G, Machuca-Gayet I, Srutkova D, Hermanova P, Martino ME, Balmand S, Hudcovic T, Heddi A et al
Lactobacillus plantarum strain maintains growth of infant mice during chronic undernutrition..
Science 2016; 351, 6275: 854-857) . Using monocolonized mouse model we showed that lactobacilli previously selected in Drosophila model of chronic undernutrition for their growth promoting capabilities were sufficient to increase linear growth of mice in a strain-dependent manner and recapitulated the whole microbiota's effect on growth. On the molecular level, we found that intestinal microbiota and Lactiplantibacillus plantarum interact with the hormonal somatotropic axis (GH/IGF-1) activity to drive systemic growth. This exciting finding implies that there might be evolutionary conserved effects of Lactiplantibacillus plantarum strains to promote juvenile growth.
 In subsequent study (Schwarzer et al., 2023Martin Schwarzer, Umesh Kumar Gautam, Kassem Makki, Anne Lambert, Tomáš Brabec, Amélie Joly, Dagmar Šrůtková, Pierre Poinsot, Tereza Novotná, Stéphanie Geoffroy, Pascal Courtin, Petra Petr Hermanová, Renata C. Matos, Jonathan J. M. Landry, Céline Gérard, Anne-Laure Bulteau, Tomáš Hudcovic, Hana Kozáková, Dominik Filipp, Marie-Pierre Chapot-Chartier, Marek Šinkora, Noël Peretti, Ivo Gomperts Boneca, Mathias Chamaillard, Hubert Vidal, Filipe De Vadder, and François Leulier
In subsequent study (Schwarzer et al., 2023Martin Schwarzer, Umesh Kumar Gautam, Kassem Makki, Anne Lambert, Tomáš Brabec, Amélie Joly, Dagmar Šrůtková, Pierre Poinsot, Tereza Novotná, Stéphanie Geoffroy, Pascal Courtin, Petra Petr Hermanová, Renata C. Matos, Jonathan J. M. Landry, Céline Gérard, Anne-Laure Bulteau, Tomáš Hudcovic, Hana Kozáková, Dominik Filipp, Marie-Pierre Chapot-Chartier, Marek Šinkora, Noël Peretti, Ivo Gomperts Boneca, Mathias Chamaillard, Hubert Vidal, Filipe De Vadder, and François Leulier
Microbe-mediated intestinal NOD2 stimulation improves linear growth of undernourished infant mice..
Science, 23 Feb 2023, Vol 379, Issue 6634) we reported that LpWJL sustains the postnatal growth of malnourished conventional animals and supports both insulin-like growth factor–1 (IGF-1) and insulin production and activity. We have identified cell walls isolated from LpWJL, as well as muramyl dipeptide and mifamurtide, as sufficient cues to stimulate animal growth despite undernutrition. Further, we found that NOD2 is necessary in intestinal epithelial cells for LpWJL-mediated IGF-1 production and for postnatal growth promotion in malnourished conventional animals. These findings indicate that, coupled with renutrition, bacteria cell walls or purified NOD2 ligands have the potential to alleviate stunting.
We are further dissecting the molecular mechanism behind the bacteria driven host growth promotion with the ultimate goal to improve the current treatment of undernutrition in human population. Further reading (Schwarzer et al., 2018Schwarzer M, Strigini M, Leulier F
Gut Microbiota and Host Juvenile Growth..
Calcified Tissue International 2018, 102(4):387-405; Schwarzer 2018Schwarzer M
Gut microbiota: puppeteer of the host juvenile growth.
Current opinion in clinical nutrition and metabolic care 2018, 21(3):179-183; Poinsot et al., 2018Poinsot P, Schwarzer M, Peretti N, Leulier F
he emerging connections between IGF1, the intestinal microbiome, Lactobacillus strains and bone growth..
Journal of Molecular Endocrinology 2018, 61(1):T103-T113) .
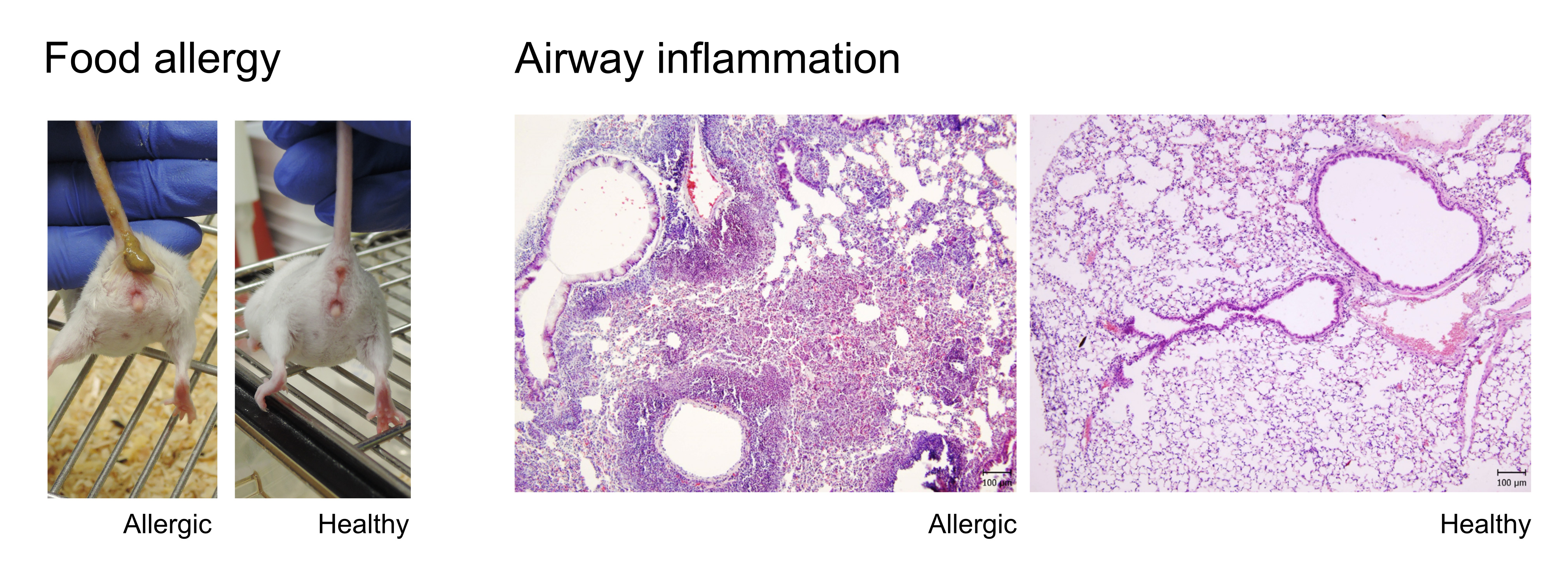
Prevention/therapy of allergic sensitization and allergy development
Allergies are widespread pathological immune reactions which are initiated by generally harmless food/airborne antigens. Their global prevalence have been increasing since the 1960s, especially in industrialized countries, suggesting environmental factors play a key role in the susceptibility and etiology of this disorder. The development of allergy is a two-step process: i) allergic sensitization and ii) the effector phase. The humoral and cellular immune responses are clearly biased toward a type 2-related phenotype, characterized by the production of specific IgE antibodies and cytokines, such as IL-4, IL-5, IL-13, or IL-10. In the effector phase, allergen-induced crosslinking of IgE bound to mast cells or basophils leads to release of allergic mediators resulting in the rapid appearance of symptoms, such as sneezing, itching, swelling or diarrhea in case of food allergies.
The hygiene hypothesis postulates that behind the increased susceptibility to allergic diseases is the lack of exposure to microbial stimuli or altered microbial stimulation, which leads to aberrant immune system maturation. Along these lines, dysbiotic microbiota leading to allergy development or associated with allergic diseases has been reported to have decreased or lack of certain groups of commensal bacteria including lactobacilli and bifidobacteria. Our group is using gnotobiotic mouse model to explore how defined bacteria consortium (Kozakova et al., 2016Kozakova H, Schwarzer M, Tuckova L, Srutkova D, Czarnowska E, Rosiak I, Hudcovic T, Schabussova I, Hermanova P, Zakostelska Z, Aleksandrzak-Piekarczyk T, Koryszewska-Baginska A, Tlaskalova-Hogenova H and Cukrowska B
Colonization of germ-free mice with a mixture of three lactobacillus strains enhances the integrity of gut mucosa and ameliorates allergic sensitization..
Cell MolImmunol 2015, doi: 10.1038/cmi.2015.09) , bacterial strains (Schwarzer et al., 2011Schwarzer M, Repa A, Daniel C, Schabussova I, Hrncir T, Pot B, Stepankova R, Hudcovic T, Pollak A, Tlaskalova-Hogenova H, Wiedermann U, Kozakova H
Neonatal colonization of mice with Lactobacillus plantarum producing the aeroallergen Bet v 1 biases towards Th1 and T-regulatory responses upon systemic sensitization..
Allergy 2011;66:368-375, Schwarzer et al., 2013Schwarzer M, Srutkova D, Schabussova I, Hudcovic T, Akgün J, Wiedermann U, Kozakova H
Neonatal colonization of germ-free mice with Bifidobacterium longum prevents allergic sensitization to major birch pollen allergen Bet v 1..
Vaccine 2013;31(46):5405-12) and defined bacterial immunogenic components (Schwarzer et al., 2016Schwarzer M, Srutkova D, Hermanova P, Leulier F, Kozakova H, and Schabussova I. Diet Matters
Endotoxin in the Diet Impacts the Level of Allergic Sensitization in Germ-Free Mice..
PLoS One 2017; 12, e0167786, Srutkova et al., 2023Dagmar Srutkova, Hana Kozakova, Tereza Novotna, Sabina Gorska, Petra Petr Hermanova, Tomas Hudcovic, Tereza Svabova, Marek Sinkora, Martin Schwarzer
Exopolysaccharide from Lacticaseibacillus rhamnosus induces IgA production in airways and alleviates allergic airway inflammation in mouse model..) impact the development of allergic sensitization and allergic inflammation.